Question 1 of 15
When sulfuric acid is added to zinc, the gas bubbles formed can be identified by?
This Acids, Bases, Salts and pH Quiz quiz contains 15 multiple choice questions designed to help you revise and test your Acids bases salts and pH quizzes knowledge. Select an answer for each question and click “Submit Answer” to see instant feedback. Take your time and try to score as high as possible!
Acids, bases and salts are very important in manufacturing and also for everyday life. They are used for the manufacture of things like soap, detergents, fertilisers, medicines and even car batteries. In this quiz, you have been provided with a question with four options and just one and only one is correct. It is important to note that these questions have been set based on the national curriculum for the gcse level.
An acid is a hydrogen-containing substance which is capable of donating a proton (which is a hydrogen ion) to another substance. On the other hand, a base is a substance which is capable of accepting the hydrogen ion from an acid. A base is characterized by its slippery texture and bitter taste. Salts are ionic compounds which result from a reaction between a base and an acid (known as a neutralization reaction). With this, salts are made up of cation and anions. pH is a measure of the acidity or basicity of a substance. The pH scale runs from 0 to 7 for acids and 7 to 14 for bases with 7 for neutral substances.
Other quizzes have been made available for here on this platform, click HERE to access them. With this, you can now go ahead and start answering the questions below.
Good luck
Question 1 of 15
When sulfuric acid is added to zinc, the gas bubbles formed can be identified by?
Question 2 of 15
Complete the word equation:
ammonia + nitric acid ==> ..???..
Question 3 of 15
In the reaction illustrated, in order to add the correct amount of acid you would add a few drops of Universal Indicator to the conical flask. If too little acid was added the indicator colour could be?
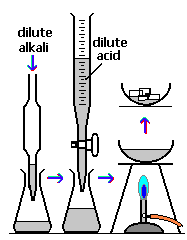
Question 4 of 15
A carbonate mineral rock was tested with dilute sulfuric acid and bubbles formed. The gas formed was?
Question 5 of 15
Which is the weakest base or alkali?
Question 6 of 15
Complete the word equation:
nickel oxide + sulfuric acid ==> ..???
Question 7 of 15
Which of the following is the pH of a strong acid?
Question 8 of 15
An iron tray was cleaned with dilute hydrochloric acid and bubbles formed. The gas formed was?
Question 9 of 15
Which of A to D completes the equation ..?..
sulfuric acid + ..?.. ==> calcium sulfate + water
Question 10 of 15
The diagram shows the preparation of a salt. Which word describes stage 4?
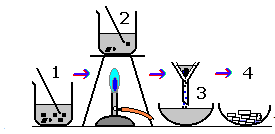
Question 11 of 15
Complete the word equation:
nickel + sulfuric acid ==> ..???..
Question 12 of 15
Insoluble bases and metals will not dissolve to form an alkaline solution, but they will dissolve in acids to form salts. This method is particularly handy for making transition metal salts. What pair of chemicals will make cobalt chloride and hydrogen?
Question 13 of 15
A small amount of a dense white powder was mixed with water in a test tube. Non of the white powder sank to the bottom of the test tube. A few drops of universal indicator were added and the solution turned blue. This meant the white powder was ...?
Question 14 of 15
Which of the following is the pH of a strong alkali?
Question 15 of 15
Which of these gives an alkaline solution when dissolved in water?